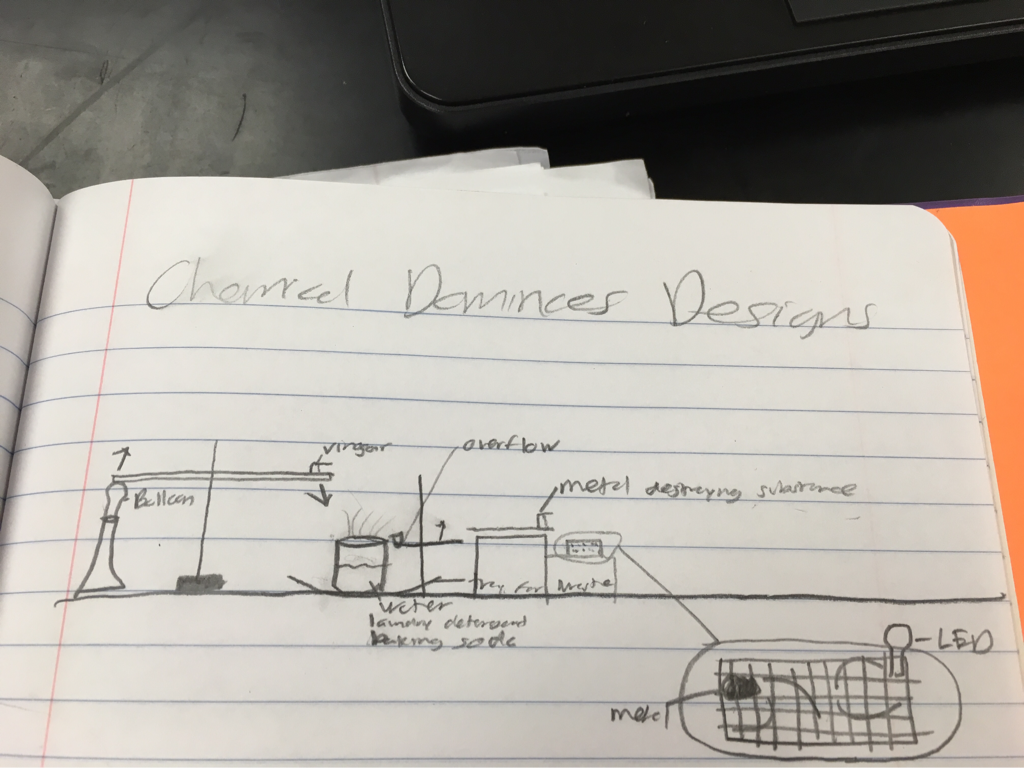
Overview - For this project we were tasked to light an LED light. To do this we set up three chemical reactions to eventually light the LED. These three reactions were vinegar and baking soda to create oxygen and raise a balloon. The second, 5mL of potassium iodide was poured into a mixture of 2mL of dish soap and 10mL to make elephant toothpaste. The third, has a tray with tin foil with electric current running through it and when the tin foil breaks the LED lights up. Before the tin foil breaks, copper chloride breaks the tin foil, making it light up.
Reflection - Overall, I feel like this project went very well for us. We were able to finish in good time, and our project did in fact work. I was in a group with Jason Franks, Kyle Jones, and Joe Malliaras. We had our difficulties being productive but in the end, we finished the job. Two peaks of this project included making a successful design and watching the product finally work. We thought our design was very good and we were proud of ourselves for making it. The two pits were both towards the very end. If you watch the video, you can see that the beaker breaks, which certainly should not have happened. Also, the LED light took awhile to come on, and I was not able to include it in the video unfortunately. However, I did greatly enjoy the project and learned a ton about chemical reactions, such as the large foaming of elephant toothpaste and how copper chloride can corrode metal.