Testing Plants
Purpose
What substances can we find from local plants that can be in potential medicines.
Materials
Balance, weight boat, lab scoops
LB broth base
Media bottles, 250 mL
Sterilizer/autoclave
Water bath, 37*C, Shaking
Sterile LB agar
Laminar flow hood and disinfectant
Safety glasses
Bunsen burner and gas lighter
Inoculating loop, Ni/Cr wire
Petri dishes, 60x15 mm, sterile
E. coli Jm109
10 mL pipet
100 mL beakers
5mm filter paper
250 mL media bottle
LB Agar
LB broth base
Plastic funnels
Procedure
Part I
1. Prepare an LB broth for the E. coli. Wait 24 hours and then add a colony of the E. coli to the broth.
2. Get six petri dishes. Draw a "+" symbol on each plate and then divide the dish into 4 quadrants. Label the quadrants 1-4 and put your initials and the date on the plate as well.
3. Liquefy LB agar by heating it in the microwave. Pour approximately 20 mL of the agar into each petri dish. Let the agar solidify for 24 hours.
Part II
1. Grind up your plant specimen in deionized water. Filter it through filter paper. Then filter the mixture again through a syringe filter. Collect 1 mL of this mixture and put it in a labeled test tube.
2. Grind up your plant specimen in methanol. Filter it through filter paper. Then filter the mixture again through a syringe filter. Collect 1 mL of this mixture and put it in a labeled test tube. Then place the test tube in a 65 degree Celsius heat block with the cap open for 24 hours. After, put 10 mL of deionized water into the test tube.
3. Repeat steps 2 and 3 until you have 6 total samples. You will have 3 of each sample.
4. Drop 3 filter paper disks into each test tube.
5. Prepare 6 negative control disks of methanol and distilled water. (3 each)
6. Prepare 6 positive control disks of ampicillin.
7. Close tubes and store at 4 degrees Celsuis.
Part III
1. Use sterile pipette to transfer 1 mL of E. coli culture to petri disc (divided in 4 quadrants)
2. Spread with flame sterilized glass spreader around disc
3. Cover and let soak for 15 minutes
4. Use sterile forceps to place discs on plates in each quadrant
5. Incubate at 37 degrees Celsius for 24 hours
Purpose
What substances can we find from local plants that can be in potential medicines.
Materials
Balance, weight boat, lab scoops
LB broth base
Media bottles, 250 mL
Sterilizer/autoclave
Water bath, 37*C, Shaking
Sterile LB agar
Laminar flow hood and disinfectant
Safety glasses
Bunsen burner and gas lighter
Inoculating loop, Ni/Cr wire
Petri dishes, 60x15 mm, sterile
E. coli Jm109
10 mL pipet
100 mL beakers
5mm filter paper
250 mL media bottle
LB Agar
LB broth base
Plastic funnels
Procedure
Part I
1. Prepare an LB broth for the E. coli. Wait 24 hours and then add a colony of the E. coli to the broth.
2. Get six petri dishes. Draw a "+" symbol on each plate and then divide the dish into 4 quadrants. Label the quadrants 1-4 and put your initials and the date on the plate as well.
3. Liquefy LB agar by heating it in the microwave. Pour approximately 20 mL of the agar into each petri dish. Let the agar solidify for 24 hours.
Part II
1. Grind up your plant specimen in deionized water. Filter it through filter paper. Then filter the mixture again through a syringe filter. Collect 1 mL of this mixture and put it in a labeled test tube.
2. Grind up your plant specimen in methanol. Filter it through filter paper. Then filter the mixture again through a syringe filter. Collect 1 mL of this mixture and put it in a labeled test tube. Then place the test tube in a 65 degree Celsius heat block with the cap open for 24 hours. After, put 10 mL of deionized water into the test tube.
3. Repeat steps 2 and 3 until you have 6 total samples. You will have 3 of each sample.
4. Drop 3 filter paper disks into each test tube.
5. Prepare 6 negative control disks of methanol and distilled water. (3 each)
6. Prepare 6 positive control disks of ampicillin.
7. Close tubes and store at 4 degrees Celsuis.
Part III
1. Use sterile pipette to transfer 1 mL of E. coli culture to petri disc (divided in 4 quadrants)
2. Spread with flame sterilized glass spreader around disc
3. Cover and let soak for 15 minutes
4. Use sterile forceps to place discs on plates in each quadrant
5. Incubate at 37 degrees Celsius for 24 hours
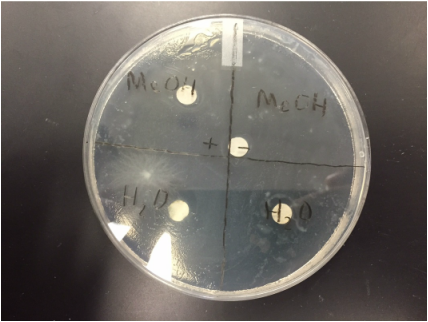
1. The positive control methane turned out to be negative
2. The negative control methane turned out to be negative
3. Both H2O control samples were positive with a .5 cm ring around it.
Analysis/Conslusion
1. My extract gave me positive results in both of the H2O controls
2. The positive methane reacted as negative and the negative H2O recorded as positive
3. Some of the filters smelled like alcohol which would jeopardize the entire experiment because the alcohol would kill any bacteria around the filters carrying the positive and negative results. Also if the pipettes that we were using was not switched with each use it would vary the result of the E. coli and hereford the result of the positive and negative controls.
4. If we could use chromatography to improve the clarity of the filters it would give clearer results.
5. If I were to carry out a different experiment I might test under different conditions, and maybe on a different bacteria. If I were to test on a different bacteria I would use the same steps except spread a different bacteria on the petri dishes near the end of the experiment. If I were to test in different condition I might incubate at different temperatures, and store at different temperatures.
Thinking Like a Biotechnician Questions:
1. If an extract gives a negative result in the antimicrobial assay, it is possible that the extract is still an antimicrobial agent, but just not for that particular bacteria. Further tests could determine if the extract has any antimicrobial properties.
2. In preparing the sample disks, some of the methanol extractions smelled like alcohol. This could be a problem because the presence of alcohol could affect the bacterial growth.
3. To identify the exact compound in an extract that causes the antimicrobial action in our plant extracts, we could separate individual components using chemical reactions and then test each of them to figure out which causes the reaction. It is possible that when multiple parts are needed for the effect, so you may need to try combinations of the separated components.
2. The negative control methane turned out to be negative
3. Both H2O control samples were positive with a .5 cm ring around it.
Analysis/Conslusion
1. My extract gave me positive results in both of the H2O controls
2. The positive methane reacted as negative and the negative H2O recorded as positive
3. Some of the filters smelled like alcohol which would jeopardize the entire experiment because the alcohol would kill any bacteria around the filters carrying the positive and negative results. Also if the pipettes that we were using was not switched with each use it would vary the result of the E. coli and hereford the result of the positive and negative controls.
4. If we could use chromatography to improve the clarity of the filters it would give clearer results.
5. If I were to carry out a different experiment I might test under different conditions, and maybe on a different bacteria. If I were to test on a different bacteria I would use the same steps except spread a different bacteria on the petri dishes near the end of the experiment. If I were to test in different condition I might incubate at different temperatures, and store at different temperatures.
Thinking Like a Biotechnician Questions:
1. If an extract gives a negative result in the antimicrobial assay, it is possible that the extract is still an antimicrobial agent, but just not for that particular bacteria. Further tests could determine if the extract has any antimicrobial properties.
2. In preparing the sample disks, some of the methanol extractions smelled like alcohol. This could be a problem because the presence of alcohol could affect the bacterial growth.
3. To identify the exact compound in an extract that causes the antimicrobial action in our plant extracts, we could separate individual components using chemical reactions and then test each of them to figure out which causes the reaction. It is possible that when multiple parts are needed for the effect, so you may need to try combinations of the separated components.