RFP Lab
Materials
2A) Materials and procedure can be found in Amgen lab manual part 2A
4A) Materials and procedure can be found in Amgen lab manual part 4A
5A) Materials and procedure can be found in Amgen lab manual part 5A
6A) Materials and procedure can be found in Amgen lab manual part 6A
Purpose
Make RFP from jelly fish in bacteria and learn about steps of genetic engineering.
Experimental Process
2A) Verification of plasmid by restriction digest
-Cut plasmid with BanH I and Hind III to cut out RFP-ara from bacterial plasmid
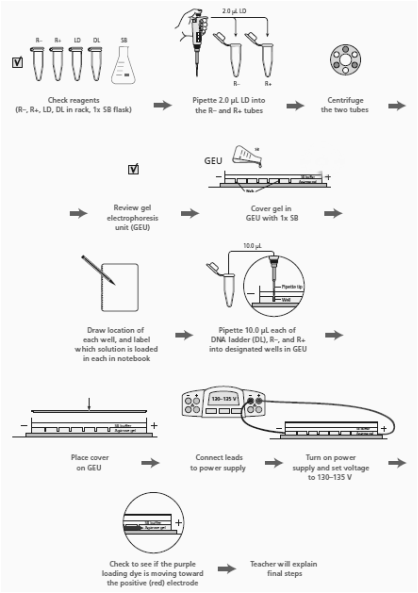
4A) Verification of the recombinant plasmid through electrophoresis
5A) Transformation of bacteria with recombinant plasmid
6A) Purification of RFP using chromotography
Results/Questions
Before 2A:
2A Questions:
Before 4A Questions:
Tubes
R+
Fragments (in order of increasing base pair size)
pARA-R: 5,302 BP
Can have all three combinations
Supercoiled configuration is fastest
Nicked circle is the medium
multimer is the slowest
R-
RFP-pBAD: 807 BP
Amp-R,Ori, Ara-C: 4,495 BP
3. Read through the Methods section on pages 64 through 66 and briefly outline the steps, using words and a flowchart.
Materials
2A) Materials and procedure can be found in Amgen lab manual part 2A
4A) Materials and procedure can be found in Amgen lab manual part 4A
5A) Materials and procedure can be found in Amgen lab manual part 5A
6A) Materials and procedure can be found in Amgen lab manual part 6A
Purpose
Make RFP from jelly fish in bacteria and learn about steps of genetic engineering.
Experimental Process
2A) Verification of plasmid by restriction digest
-Cut plasmid with BanH I and Hind III to cut out RFP-ara from bacterial plasmid
4A) Verification of the recombinant plasmid through electrophoresis
5A) Transformation of bacteria with recombinant plasmid
6A) Purification of RFP using chromotography
Results/Questions
Before 2A:
- If pARA-R is digested with BamHI and HindIII, what fragments are produced? Record the nucleotide sequence of the sticky ends and the length of each fragment (bp), and indicate the genes and other important sequences present on each fragment.
-Two fragments are produced; two RFP fragments one with pBAD and one with Ara-C with ori with Amp-R. The RFP and pBAD is 807 BP, while Ara-C, ori, and Amp-R is 4495 BP. - In order to create a plasmid that can produce the red fluorescent protein in bacteria, what components are needed in the plasmid?
-You need the RFP gene and the promoter, Ara-C. - If the uptake of DNA by bacteria is inefficient (as discussed in the reading), why is a selectable marker (gene with resistance to an antibiotic) critical in cloning a gene in bacteria?
-The marker is critical because it allows us to control how much bacteria will grow and live, while the other bacteria will die. This separates the bacteria we need, the ones we added the gene to, and those we don't need, the ones that were not altered.
2A Questions:
- List in words or indicate in a drawing the important features of a plasmid vector that are required to clone a gene. Explain the purpose of each feature.
-Ori- origin of replication , RFP- red fluorescent protein: our gene of interest, Amp-R: selective marker, Ara-C: protein(transcription factor) binds to promoter in order to get transcription of our gene of interest - What role do restriction enzymes have in nature?
-They are used as defense mechanisms. By cutting out DNA of other bacteria they die giving bacteria a better chance of survival. - Using your understanding of evolution, why would bacteria retain a gene that gives them resistance to antibiotics? How is the existence of bacteria with antibiotic resistance affecting medicine today?
-This allows the bacteria not die off due to antibiotics, and pass it on to later generations creating more bacteria with the gene. - Bacteria, sea anemones, and humans seem, on the surface, to be very different organisms. Explain how a gene from humans or a sea anemone can be expressed in bacteria to make a product never before made in bacteria.
-All cells follow the same central dogma and process of production of protein. This means bacteria and human cells make protein in the same way,DNA to mRNA through RNA polymerase, and mRNA to protein through transcription. - Due to a mishap in the lab, bacteria carrying a plasmid with an ampicillin resistant gene and bacteria carrying a plasmid with a gene that provides resistance to another antibiotic (kanamycin) were accidentally mixed together. Design an experiment that will allow you to sort out the two kinds of bacteria.
-You take two petri dishes and put Kan on one, so the Kan resistant will live. On the other petri dish Amp, so the Amp resistant will live. Now you have two dishes with separated bacteria.
Before 4A Questions:
- The pARA-R plasmid you digested in Laboratory 2A was replicated in a bacterial cell. What configurations—supercoiled, nicked circle, and multimer—might the plasmid have before digestion?
-The plasmid can have all three configurations: supercoiled, nicked circle, and multimer. - You need to catalog all the products you might see, including the different plasmid configurations. Review your work in Laboratory 2A. What products might you expect to see in the R– and R+ tubes? Create a table that shows all the possible fragments and plasmids by tube. Include the length (bp size) of each possible fragment or plasmid, and arrange the products found in each microfuge tube by size, from smallest to largest. Include any possible plasmid configuations, and arrange them first by size and next by speed through the gel, from fastest to slowest.
Tubes
R+
Fragments (in order of increasing base pair size)
pARA-R: 5,302 BP
Can have all three combinations
Supercoiled configuration is fastest
Nicked circle is the medium
multimer is the slowest
R-
RFP-pBAD: 807 BP
Amp-R,Ori, Ara-C: 4,495 BP
3. Read through the Methods section on pages 64 through 66 and briefly outline the steps, using words and a flowchart.
4A Questions:
1. Why is it important to verify that you have the correct recombinant plasmid?
-It is important to verify because during the lab, because we could make measuring mistakes, the materials could be faulty, or make errors in our procedure.
2. How did your actual gel results compare to your gel predictions?
-We predicted that all bacteria will go on the LB plate, on the LB-AMP plate P- won't grow and P+ will grow. On the ARA plate only the red bacteria will grow.
3. Do you see any bands that are not expected? What could explain the origin of these unexpected bands?
-Our group had no unexpected bands. but we did not expect there to be missing bands.
Before Lab 5A Questions:
1. Ampicillin is an antibiotic that kills bacterial cells by disrupting the formation of cell walls. However, the pARA-R plasmid has the ampicillin resistance gene, which produces a protein that breaks down ampicillin. What is the purpose of growing bacteria that have been transformed in the presence of ampicillin?
-The purpose is separation of those with the resistance gene and those without.
2. What will happen when bacterial cells that contain the pARA-R plasmid are not given arabinose?
-When they are without the arabinose the RFP gene will not be expressed and there will be no red glow.
3. In the lab, you will add samples of the control group P– and the treatment group P+ to plates that contain various combinations of Luria Broth (LB), ampicillin, and the sugar arabinose. Predict the growth on each dish.
-I predict that Plate I (LB) will have non-glowing growth on both sides. I predict that Plate II (LB/AMP) will have non-glowing growth on the P+ side. For Plate III (LB/AMP/ARA), I predict a glowing red colony.
5A Questions:
1. Look at the results of your transformation. Do your actual results match your predicted results? If not, what differences do you see, and what are some explanations for these differences?
-Our predictions were right except there was no red glowing on the LB/AMP/ARA plate. Glowing on that plate was the predicted result, but it did not occur.
2. How many red colonies were present on your LB/AMP/ARA plate?
-There were no red colonies that appeared on the plate.
3. Why did the red colonies only appear on the LB/AMP/ARA plate and not the LB/Amp plate?
-They need ARA-C in order for transcription to occur and gene expression.
4. Recombinant plasmids are engineered so that they can replicate in the cell independently of the chromosome replication. Why is it important to have multiple copies of a recombinant plasmid within a cell?
-It is so there is extra for cell growth and in case of failure during transcription.
5. How is the information encoded in the RFP gene expressed as a trait? Be sure to use what you have previously learned about gene expression and the relationship between DNA, RNA, protein, and traits.
-The expression happen when the DNA is transcribed into protein using the central dogma DNA to mRNA to Protein.
6. Why is it possible for bacteria to make a human protein, such as insulin, or a sea anemone protein, such as the red fluorescent dye?
-All genes have the same central dogma (DNA to mRNA to Protein), so they can be expressed in seemingly different organisms.
6 Questions:
1. How can solutions of different salt concentrations, which will unfold proteins to varying degrees, be used to help purify red fluorescent protein using column chromatography? Read through the Methods sections for Part A and for Part B and briefly outline the steps, using words and a flowchart.
-It determines the promoter regions of each protein. These different regions give each protein different functions.
2. What properties of the amino acids in a protein relate to protein folding?
-The sequence of amino acids determines it folds into a protein.
3. Does the eluate containing your red fluorescent protein appear less bright or brighter than it did in the cell lysate following centrifugation? If there is a noticeable difference in the intensity of the red color, what might account for that?
-The RFP-containting eluate is brighter than the cell-lysate, because the elute contains the most RFP,while the cell lysate contained all the cells.
4. What characteristic of red fluorescent protein is used as the basis for separation by column chromatography?
-The red florescent protein attached to the resin column when unfolded.
5. How might the column chromatography procedure be adjusted or modified to increase the purity of the red fluorescent protein sample?
-If we repeated the experiment or lab with more wash buffers, and were more careful about collection, we would increase the expression of the RFP and we would see the protein more.
1. Why is it important to verify that you have the correct recombinant plasmid?
-It is important to verify because during the lab, because we could make measuring mistakes, the materials could be faulty, or make errors in our procedure.
2. How did your actual gel results compare to your gel predictions?
-We predicted that all bacteria will go on the LB plate, on the LB-AMP plate P- won't grow and P+ will grow. On the ARA plate only the red bacteria will grow.
3. Do you see any bands that are not expected? What could explain the origin of these unexpected bands?
-Our group had no unexpected bands. but we did not expect there to be missing bands.
Before Lab 5A Questions:
1. Ampicillin is an antibiotic that kills bacterial cells by disrupting the formation of cell walls. However, the pARA-R plasmid has the ampicillin resistance gene, which produces a protein that breaks down ampicillin. What is the purpose of growing bacteria that have been transformed in the presence of ampicillin?
-The purpose is separation of those with the resistance gene and those without.
2. What will happen when bacterial cells that contain the pARA-R plasmid are not given arabinose?
-When they are without the arabinose the RFP gene will not be expressed and there will be no red glow.
3. In the lab, you will add samples of the control group P– and the treatment group P+ to plates that contain various combinations of Luria Broth (LB), ampicillin, and the sugar arabinose. Predict the growth on each dish.
-I predict that Plate I (LB) will have non-glowing growth on both sides. I predict that Plate II (LB/AMP) will have non-glowing growth on the P+ side. For Plate III (LB/AMP/ARA), I predict a glowing red colony.
5A Questions:
1. Look at the results of your transformation. Do your actual results match your predicted results? If not, what differences do you see, and what are some explanations for these differences?
-Our predictions were right except there was no red glowing on the LB/AMP/ARA plate. Glowing on that plate was the predicted result, but it did not occur.
2. How many red colonies were present on your LB/AMP/ARA plate?
-There were no red colonies that appeared on the plate.
3. Why did the red colonies only appear on the LB/AMP/ARA plate and not the LB/Amp plate?
-They need ARA-C in order for transcription to occur and gene expression.
4. Recombinant plasmids are engineered so that they can replicate in the cell independently of the chromosome replication. Why is it important to have multiple copies of a recombinant plasmid within a cell?
-It is so there is extra for cell growth and in case of failure during transcription.
5. How is the information encoded in the RFP gene expressed as a trait? Be sure to use what you have previously learned about gene expression and the relationship between DNA, RNA, protein, and traits.
-The expression happen when the DNA is transcribed into protein using the central dogma DNA to mRNA to Protein.
6. Why is it possible for bacteria to make a human protein, such as insulin, or a sea anemone protein, such as the red fluorescent dye?
-All genes have the same central dogma (DNA to mRNA to Protein), so they can be expressed in seemingly different organisms.
6 Questions:
1. How can solutions of different salt concentrations, which will unfold proteins to varying degrees, be used to help purify red fluorescent protein using column chromatography? Read through the Methods sections for Part A and for Part B and briefly outline the steps, using words and a flowchart.
-It determines the promoter regions of each protein. These different regions give each protein different functions.
2. What properties of the amino acids in a protein relate to protein folding?
-The sequence of amino acids determines it folds into a protein.
3. Does the eluate containing your red fluorescent protein appear less bright or brighter than it did in the cell lysate following centrifugation? If there is a noticeable difference in the intensity of the red color, what might account for that?
-The RFP-containting eluate is brighter than the cell-lysate, because the elute contains the most RFP,while the cell lysate contained all the cells.
4. What characteristic of red fluorescent protein is used as the basis for separation by column chromatography?
-The red florescent protein attached to the resin column when unfolded.
5. How might the column chromatography procedure be adjusted or modified to increase the purity of the red fluorescent protein sample?
-If we repeated the experiment or lab with more wash buffers, and were more careful about collection, we would increase the expression of the RFP and we would see the protein more.
|
Our column was the purple line on the first gel which indicates that we had very little protein in the proper places and our gel had some issues as seen in the odd structure or formation. However, compared to the other classes we saw that some groups had better RFP extraction and the gel showed it to be more pure than others. They had thicker bands at places you would expect the bands to be at.
|
Analysis and conclusion
During the lab we attempted to create a colony of bacteria that glowed red using RFP gene. The colony we grew did not glow red, however we did successfully grow a colony with the RFP gene in it, but the gene was never expressed. We did this through the use of restriction enzymes to isolate the RFP gene, and obtained conformation that it was the right gene using electrophoresis. Next we injected bacteria with the gene, and grew them on a petri dish.Lastly we used recombinant plasmids and chromatography to separate the protein into a tube.
Reflection
I think our group worked well together. We were very quick and efficient during the lab which helped me start on other assignments for the class. The only regret I had was that Poh used on of the machine protein separator pumps and that wasted a day and the machine ultimately amounted to no progress in the day.
During the lab we attempted to create a colony of bacteria that glowed red using RFP gene. The colony we grew did not glow red, however we did successfully grow a colony with the RFP gene in it, but the gene was never expressed. We did this through the use of restriction enzymes to isolate the RFP gene, and obtained conformation that it was the right gene using electrophoresis. Next we injected bacteria with the gene, and grew them on a petri dish.Lastly we used recombinant plasmids and chromatography to separate the protein into a tube.
Reflection
I think our group worked well together. We were very quick and efficient during the lab which helped me start on other assignments for the class. The only regret I had was that Poh used on of the machine protein separator pumps and that wasted a day and the machine ultimately amounted to no progress in the day.